Q1. Which of the following metal doesn't react with water as well as with steam?
Solution
Lead does not react with water (or even steam).
Q2. What products are formed when aluminum reacts with dilute sulphuric acid?
Solution
Aluminum reacts with dilute sulphuric acid to form aluminium sulphate with the liberation of hydrogen gas.
2Al + 3H2SO4 → Al2(SO4)3+ 3H2 ↑
Q3. The green coating on copper vessel is a mixture of __________ and copper hydroxide.
Solution
The green coating on copper vessel is a mixture of copper carbonate and copper hydroxide.
Q4. Which is a property of an acidic oxide?
Solution
Acidic oxides turns moist blue litmus paper to red being acidic in nature.
Q5. Which gas is evolved when carbon reacts with dilute sulphuric acid?
Solution
Carbon is a non-metal and non-metals do not react with acids.
Q6. Which of the following metals can be easily cut with a knife?
Solution
Sodium is a metal that is very soft and can be easily cut with a knife.
Q7. Which of the following represents a displacement reaction?
Solution
Those reactions in which one element takes the place of another element in a compound are known as displacement reaction.
In reaction,
A + BC → AC + B
A takes the place of element B in its compound BC to form product AC.
Q8. How do you know that a substance is a metal?
Solution
Metals are substances that are good conductors of heat and electricity.
Q9. What does this reaction suggest?MgSO4 + Cu  CuSO4 + Mg
CuSO4 + Mg
Solution
Metal that are less reactive, cannot displace more reactive metal from their salt solution. Hence the reaction will not occur.
Q10. Tungsten is used in electric bulbs because of its:
Solution
Tungsten is used in electric bulbs because it is metallic in nature and hence conducts electricity.
Q11. The property that allows metals to be hit and hammered into thin sheets is:
Solution
The property that allows metals to be hit and hammered into thin sheets is malleability.
Q12. Select the pair that shows similar type of reaction with air and water.
Solution
Both Copper (Cu) and Calcium (Ca) are metals. They react with air and water to produce compound that is basic in nature.
Q13. We use aluminium foils for wrapping food. This shows that aluminium is:
Solution
Metals can be beaten to thin sheets. This property is known as malleability.
Q14. What happens when zinc is added to a solution of iron sulphate?
Solution
Zinc is more reactive than iron. Therefore, when zinc is added to the solution of iron sulphate, the colour of iron sulphate solution changes and a grey precipitate of iron and a colourless solution of zinc sulphate is formed.
Q15. _______ are used to make strings of musical instruments.
Solution
Metals are used to make strings of musical instruments.
Q16. (i) What happens when potassium comes in contact with water? (ii) Write chemical reaction involved.
Solution
(i) Potassium reacts vigorously with cold water forming potassium hydroxide (KOH) and hydrogen (H2). The reaction of potassium metal with water is highly heat producing due to which the hydrogen gas formed during the reaction catches fire immediately. (ii) 2K + 2H2O  2KOH + H2 + Heat
2KOH + H2 + Heat
Q17. Carbon dioxide dissolves in water to form _______ acid.
Solution
Carbon dioxide dissolves in water to form carbonic acid.
Q18. Metal detectors work on the principle of conduction of electricity by metals.
Solution
True
Q19. Generally, non metals are not lustrous. Which of the following non-metal is lustrous?
Solution
Generally, non metals are not lustrous but iodine being an exception is lustrous.
Q20. When sulphur dioxide dissolves in water, it produces:
Solution
Non-metal oxide on reaction with water produces acidic solution.
Q21. Name a metal which is used in making mirrors. Which property makes it suitable for this?
Solution
Silver metal is used in making mirrors. Due to shiny appearance silver metal is an excellent reflector of light.
Q22. Why are metals used for making electric cables?
Solution
Metals are good conductors of electricity and are ductile i.e. can be drawn into thin wires hence they are used for making wires.
Q23. Which of the following reaction will not occur?
Solution
Copper is less reactive than zinc so it cannot displace zinc from its solution. Therefore, this reaction will not occur.
Q24. Which of the following material will heat up fast?
Solution
As metals are good conductors of heat, they will heat up fast.
Q25. Which of the following metal is present in blood?
Solution
Iron is the metal present in blood hemoglobin.
Q26. A screw-driver is provided with wooden handle as it is not slippery.
Solution
False.
A screw-driver is provided with wooden handle as it is slippery.
Q27. What is meant by saying that metals are: (i) malleable (ii) ductile (iii) lustrous (iv) conductors, and (v) sonorous?
Give one application or use of each property.
Solution
(i) Malleable: This means that metals can be beaten into thin sheets with a hammer. The property which allows the metals to be hammered into thin sheets is called malleability.
Aluminium metal can be hammered to form alumimium foils.
(ii) Ductile: This means that metals can be drawn (or stretched) into thin wires. The property which allows the metals to be drawn into thin wires is called ductility.
Copper metal can be drawn into thin copper wires (used as electric wires).
(iii) Lustrous: This means that metals have a shiny appearance. The shiny appearance of the metals is called metallic lustre.
Gold metal is used for making jewellery because of its shiny lustre.
(iv) Conductors: This means that metals allow heat and electricity to pass through them easily.
Copper metal is used in making cooking utensils because of its high heat conductivity.
(v) Sonorous: This means that metals make a ringing sound when we strike them. The property by virtue of which metals make a ringing sound is called sonority.
Metal sheets are used for making bicycle bells and temple bells.
Q28. Coal is malleable.
Solution
False.
Coal is non metal hence it does not show malleability
Q29. All metals react equally with water and air.
Solution
False.
Some metals react with water and air.
Q30. What products are formed when iron (Fe) reacts with dilute hydrochloric acid (HCl)?
Solution
Fe + 2HCl 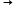 FeCl2 + H2
FeCl2 + H2
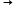 FeCl2 + H2
FeCl2 + H2
Q31. Oxide of magnesium is basic in nature because:
Solution
Oxide of magnesium is basic in nature because when magnesium oxide is dissolved in water then the resulting solution is basic in nature due to the presence of Mg(OH)2 which turns red litmus to blue.
MgO + H2O  Mg(OH)2
Mg(OH)2
 Mg(OH)2
Mg(OH)2
Q32. Metals react with water to form either their respective oxides or hydroxides and liberate:
Solution
Metals react with water to form either their respective oxides or hydroxides and liberate hydrogen gas.
Q33. (i) Name the metal which is used for decorating sweets and which property makes it suitable for this? (ii) Write another use of this metal.
Solution
(i) Silver foils are used for decorating sweets. It is because of its high malleability, silver metal can be hammered into thin silver sheets. (ii) Silver metal is used for making jewellery.
Q34. The metal which is used in constructing railway lines and girders is:
Solution
Iron is used in constructing railway lines and girders because it is a very strong metal.
Q35. Select a basic oxide.
Solution
Metals on reaction with oxygen produce basic oxides.
Q36. Sodium catches fire when it comes in contact with water because:
Solution
Sodium reacts vigorously with cold water forming sodium hydroxide (NaOH) and hydrogen(H2). The reaction of sodium metal with water is highly heat producing due to which the hydrogen gas formed during the reaction catches fire and burns causing little explosions.
Q37. The substances calcium, copper and aluminum, all are _______.
Solution
The substances calcium, copper and aluminum, all are metals.
Q38. (i)CuSO4 + Fe → FeSO4 + Cu
(ii)FeSO4 + Zn → ZnSO4 + Fe
Which of the following statement is true on the basis of above reactions?
Solution
In the first reaction, Fe displaces Cu. Therefore, Fe is more reactive than Cu (reactivity of Fe > reactivity of Cu).
In the second reaction Fe is displaced by Zn. Therefore, Fe is less reactive than Zn (reactivity of Zn > reactivity of Fe).
Therefore, Zn is more reactive and Cu is least reactive.
Q39. Hydrochloric acid turns:
Solution
Acids turn blue litmus solution red.
Q40. The metal which reacts with water only when it is red hot is:
Solution
Iron reacts with water only when it is red hot and steam is passed over it.
3Fe + 4H2O(steam)  Fe3O4 + 4H2
Fe3O4 + 4H2
 Fe3O4 + 4H2
Fe3O4 + 4H2
Q41. Rust of iron is basically:
Solution
Iron reacts with water and oxygen to produce rust which is an oxide.
The reaction is as follows:
4Fe + 3O2 + 2H2O → 2Fe2O3.2H2O
Iron rusts in moist air to form hydrated ferric oxide.
Q42. Metals are more important to human life than non-metals.
Solution
False.
Metals as well as non metals are important to human life.
Q43. Considering the following reactions, which is the most reactive metal of the given metals?Mg + ZnSO4 → MgSO4 +Zn2Na + MgSO4 → Na2SO4 + Mg2Na + ZnSO4 → Na2SO4 +Zn
Solution
This is since both Na and Mg are displacing zinc from zinc sulphate solution, so both are more reactive than zinc. But Na is displacing Mg from its salt solution. Hence, Na is more reactive than Mg.
Q44. Metals can be easily beaten into sheets, this property is called _______.
Solution
Metals can be easily beaten into sheets, this property is called malleability.
Q45. _______ gas burns with a pop sound.
Solution
Hydrogen gas burns with a pop sound.
Q46. Which of the following is the only liquid metal?
Solution
Mercury is the only metal which is liquid at room temperature.
Q47. From the given three equations, which metal will be the most reactive out of the given metals? Zn + PbSO4  ZnSO4 + PbFe + CuSO4
ZnSO4 + PbFe + CuSO4  FeSO4 + CuFe + ZnSO4
FeSO4 + CuFe + ZnSO4  FeSO4 + Zn
FeSO4 + Zn
Solution
In 1st equation Zn displaces Pb. But in 2nd and 3rd chemical equations, Fe displaces both Cu and Zn. So, Fe is the most reactive.
Q48. Select the correct pair of metals that cannot be stored in air.
Solution
Both sodium and Potassium are very reactive metals. They catch fire when exposed to air, therefore are stored in kerosene.
Q49. Metals are generally good thermal and electrical conductors.
Solution
True
Q50. Why is copper a good metal to use for electrical wires?
Solution
Copper is a good conductor of electricity.
Q51. Which physical property of metals is shown in following picture of a shining spoon, knife and fork?

Solution
The shining spoon, knife and fork are made of metals. This shows that metals have shiny lustre.
Q52. What happens when a piece of copper metal is placed in iron sulphate solution?
Solution
When a piece of copper is placed in iron sulphate solution then no change will be observed. This is because copper is less reactive than iron, therefore, unable to displace it from its solution.
Q53. Aluminium is light in weight and a good conductor of heat.
Solution
True
Q54. Which of the following is TRUE about metals?
Solution
Metals allow heat to pass through them. Therefore, they are good conductors of heat.
Q55. Write the equations for the reactions of:
(i) Iron with dilute hydrochloric acid.
(ii) Aluminium with dilute hydrochloric acid
(iii) Gold with dilute hydrochloric acid
Solution
(i) Fe + 2HCl  FeCl2 + H2
(ii) Al + 6HCl
FeCl2 + H2
(ii) Al + 6HCl  AlCl3 + 3H2
(iii) Au + HCl
AlCl3 + 3H2
(iii) Au + HCl  no reaction
no reaction
Q56. Name: (a) A metal which is liquid at room temperature. (b) Two metals which are soft and can be cut with a knife.
Solution
(a) A metal which is liquid at room temperature :- Mercury (b) Two metals which are soft and can be cut with a knife :- Sodium and potassium
Q57. Gold and silver are most malleable metals.
Solution
True
Q58. (i) Give name of two metals which are used for making frying pans and geysers. Which property makes them suitable for making utensils and water boilers? (ii) Name a metal which is a poor conductor of heat.
Solution
(i) Copper and aluminium are used for making frying pans and geysers because they are very good conductors of heat. (ii) Lead is a poor conductor of heat.
Q59. The metals which are usually used for making cooking utensils and water boilers are:
Solution
Copper and aluminium are used for making cooking utensils and water boilers because they are very good conductors of heat.
Q60. Which of the following is used in cookware?
Solution
Cookwares are made of metals as they are good conductors of heat.
Q61. (i) Why does the colour of copper sulphate solution change when an iron nail is kept immersed in it? What is the change in colour observed? (ii) What happens to the iron metal?
Solution
(i) Iron metal is more reactive than copper. Therefore, it displaces copper from copper sulphate solution (CuSO4). The deep blue colour of copper sulphate solution fades due to the formation of light green solution of iron sulphate. (ii) A red-brown coating of copper metal is formed on the surface of iron nail.
Q62. The property of metals by which they can be drawn into wires is called ________.
Solution
The property of metals by which they can be drawn into wires is called ductility.
Q63. Non-metals are found as solids, liquids or gases.
Solution
True
Q64. Metals are ________ conductors of electricity.
Solution
Metals are good conductors of electricity.
Q65. The property that allows heat and electricity to pass through metals easily is:
Solution
Metals are good conductors of heat and electricity as they allow it to pass through them.
Q66. Sodium metal is kept immersed in kerosene and not water because:
Solution
Sodium is a highly reactive metal and reacts violently with oxygen and water producing a large amount of heat and light.
Q67. (i) Why does copper coin dissolve in the solution of silver nitrate if it is immersed in it for some time? (ii) What colour change is observed?
Solution
(i) Copper is more reactive than silver. Therefore, when copper coin is kept immersed in a solution of silver nitrate, it will displace silver from silver nitrate solution and a solution of copper nitrate will be formed. Thus, copper coin will dissolve in the solution. (ii) The colour of the solution will change from colourless to blue.
Q68. (i) Fe + CuSO4  FeSO4 + Cu (ii) Zn + FeSO4
FeSO4 + Cu (ii) Zn + FeSO4  ZnSO4 + Fe On the basis of the above reactions, indicate which is most reactive and which is least reactive metal out of zinc, copper, and iron.
ZnSO4 + Fe On the basis of the above reactions, indicate which is most reactive and which is least reactive metal out of zinc, copper, and iron.
 FeSO4 + Cu (ii) Zn + FeSO4
FeSO4 + Cu (ii) Zn + FeSO4  ZnSO4 + Fe On the basis of the above reactions, indicate which is most reactive and which is least reactive metal out of zinc, copper, and iron.
ZnSO4 + Fe On the basis of the above reactions, indicate which is most reactive and which is least reactive metal out of zinc, copper, and iron.Solution
In the first reaction, Fe displaces Cu. Therefore, Fe is more reactive than Cu (reactivity of Fe> reactivity of Cu). In the second reaction Fe is displaced by Zn. Therefore, Fe is less reactive than Zn (reactivity of Zn> reactivity of Fe). Therefore, Zn is more reactive and Cu is least reactive.
Q69. Gold metal is used for making jewellery because it only reacts with air and that too slowly.
Solution
True
Q70. All metals are solids except __________.
Solution
All metals are solids except mercury.
Q71. (i) Copper is used for making electric wires. But how do metals conduct electricity? (ii) Which metal is the best conductor of electricity?
Solution
(i) Metals offer very less resistance to the flow of electric current. They conduct electricity because of the presence of free electrons in them. These free electrons can move easily through the metal and thus conduct electricity. (ii) Silver is the best conductor of electricity.
Q72. (i)Explain, why, when a copper object remains in damp air for a considerable time, a green coating is formed on its surface. What is the chemical formula of that green colour substance? (ii) Write the chemical reaction involved.
Solution
(i) When a copper object remains in damp air for a considerable time, a green coating of copper carbonate is formed on its surface. It is formed due to the reaction of copper with carbon dioxide of air and water vapour also present in air. The chemical formula of green colour substance is CuCO3(copper carbonate). (ii) 2Cu + H2O + CO2 + O2  Cu(OH)2 + CuCO3
Cu(OH)2 + CuCO3
Q73. Which property of metals makes them suitable for use in jewellery?
Solution
Two properties of metals makes them suitable for use in jewellery: (a) Metals have a shiny lustre. (b) Metals are malleable and ductile.
Q74. Metals and water react to form a metal _______ and hydrogen gas.
Solution
Metals and water react to form a metal hydroxide and hydrogen gas.
Q75. Differentiate between metals and non-metals on the basis of their physical properties (four points).
Solution
Difference in Physical Properties of Metals and Non-Metals:
Metals
Non-metals
1. Metals are malleable and ductile.
1. Non-metals are neither malleable nor ductile.
2. Metals are good conductors of heat and electricity.
2. Non-metals are poor conductors of heat and electricity.(except graphite which is a good conductors of electricity).
3. Metals are lustrous.
3. Non-metals are non-lustrous.
4. Metals are strong .They have high tensile strength.(except sodium and potassium which are not strong and have low tensile strength).
4. Non-metals are not strong.They have low tensile strength.
Q76. A metal with silvery appearance is slowly tarnished in air. It reacts with boiling water and evolves hydrogen gas. Dilute hydrochloric acid reacts vigorously with metal evolving hydrogen gas. (i)Identify the metal and (ii) Also write the reactions involved.
Solution
(i)The metal is magnesium.
(ii)Mg + 2H2O  Mg(OH)2 + H2
Mg + 2HCl
Mg(OH)2 + H2
Mg + 2HCl  MgCl2 + H2
MgCl2 + H2
Comments
Post a Comment